This is a cancer update. First, I want to let you know what’s going on after my meetings with my oncologists and my next plans. Next I’ll do a deep dive into the kinds of evidence that supports various medical treatments. When you hear a doctor say “we don’t really have evidence for that”, what do they mean?
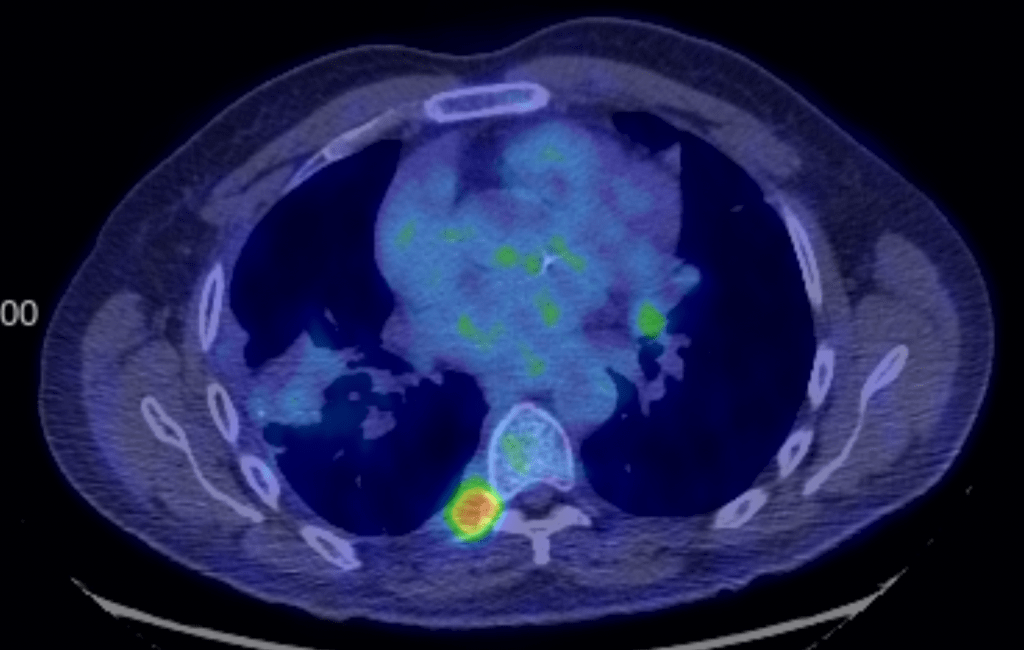
Update: My March 5th PET scan showed that while most of my tumors are completely gone, I have a few persistent tumors that will need to be addressed. I have 2 prominent ones, and a few minor ones. We met with my chemo oncologist and we agreed that the current approach isn’t quite working and we need a different approach. We decided to get a biopsy on the lung tumor which is relatively accessible. So just yesterday, we went to the hospital for a lung biopsy, performed by my pulmonologist. The procedure went well. He’ll send off the biopsy for Next Generation Sequencing which will give us information on the genetics of the tumor. This should give us good information about what chemo or immunotherapy treatments to use next. We’ll have that information back in a few weeks.
Then we spoke to my radiation oncologist just today. He’s mostly concerned about the tumor near the spine. The good news is that he thinks it’s far enough away from my spinal cord that he can treat it without doing any damage to my nervous system, which is great. That will probably happen in about 3 weeks. We’ve had success with radiation in the past, and we’re encouraged that he can treat that area.
Alternative treatments: Because we are eager to put this whole cancer thing behind us, we’ve decided to “give it both barrels” and do everything we can to end all of my tumors and move on with life. So in addition to the “traditional” medical means, we are also going to pursue some “alternative” measures. We learned about a program called “Be Resilient” from a friend. This program doesn’t claim to “cure cancer” per se, but they do seek to learn why your body is a “good host” for cancer cells, and to optimize your body’s ability to fight back. The first thing they will do is test my blood for over 400 biomarkers to see what my body chemistry is like. Then they’ll put together a plan for me to optimize my body chemistry. Then I’ll work on the program for 8 months, keeping tabs on how I’m doing on the way. We’ll also continue with the medical treatments recommended by our oncologists. Will it work? Of course I don’t really know, but at this point I feel like I need to get serious and try everything I can.
The program is not free! As I’ve mentioned before, most of my medical care is being paid for by us, and reimbursed through a cost sharing program. But our program does not reimburse for “alternative treatments”. Our friend Annely has put together a Givesendgo page for me if you’d like to contribute.
We have been so grateful for all the support we’ve gotten from all of you over the past 2 years! Please do not contribute to this fund if it will challenge you financially. We are blessed to have lots of friends!
Please continue to pray for wisdom for us and our doctors as we go through this process, and of course for God’s healing!
Evidence: This section is going to be a detailed discussion of the kinds of evidence that supports various scientific and medical findings. Read on if you find that interesting, otherwise, you can skip the rest of this post.
You may have been in your doctor’s office and been discussing some new treatment you’ve heard about. They may have said “there isn’t really any evidence to support that”, but you heard some pretty convincing stuff about it. The reason for all this is that there are different levels of evidence that may be convincing or useful in some circumstances but not others. I thought I’d discuss this a little to clarify this, especially as it pertains to “alternative treatments”. Here are some of the kinds of evidence we commonly deal with in the medical or scientific community. You also run across some of these in every day life.
Anecdotal evidence: This kind of evidence is basically a collection of stories people have from every day life. Things like “My aunt tried a supplement and it really worked for her” or “I got the vaccine and started having ringing in my ears”. This kind of evidence can and does often form the starting point for more rigorous studies, but by themselves, they have a lot of problems. Private citizens often don’t have a firm understanding of cause and effect, and usually don’t take into account all the factors that may contribute to a result. Like a joke study someone told me about in which a city determined that ice cream consumption at a local beach was correlated to shark attacks. So what does this mean? Does eating ice cream cause shark attacks? Of course not. Hot weather causes people to eat ice cream and also causes people to swim in the ocean. A third factor causes both results.
But this is not to say that anecdotal evidence is worthless. When a lot of people have similar stories, it can and does cause scientists to be interested in finding out what’s going on. Anecdotal evidence, once properly understood, can lead to real scientific findings.
Many of the ads your see on TV or the internet contain testimonials and claims that basically amount to anecdotal evidence. This is why we have to consider these kind of claims with caution. Many of the supplements and much of nutritional advice we see are not supported by the rigorous evidence I will discuss below. What they claim may be true, but we can’t know for sure just based on the evidence they present.
Case studies: It’s pretty common for doctors or scientists to have an interesting case that they write up and publish in a journal. These studies may include just 1 or a handful of patients. This represents real scientific evidence, but since the sample numbers are so low, they are often ignored or not taken very seriously by the medical or scientific communities. However, if enough of this evidence is published, they can trigger more rigorous studies.
Many of what we consider alternative treatments are based on anecdotal evidence or case studies. There is evidence for them, but the evidence is not as rigorous as we may like. This is why the medical community is very slow to promote these kinds of treatments. Unfortunately, some very useful treatments may go under-utilized because no one wants to pay for more rigorous studies to be done.
Observational studies: These studies are performed by collecting available data and trying to come up with a useful conclusion. This method is often used when it’s considered unethical, too expensive, or too time consuming to collect new data. Many papers regarding COVID represented this kind of data.
These studies can often lead to useful conclusions, but because the researchers can’t control the kind of information they gather, it can be hard to sort out what connections are real, and what are the result of other factors. A good example of this comes from COVID studies estimating the number of real cases early in the pandemic. Researchers would collect information from hospitals about the number of patients who had COVID. Then they would announce this as a percentage of the population with the disease. The problem is that the general public and the people who came to the hospital are 2 different groups. The people who came to the hospital are “self selecting” as people who are more engaged with the health care system, have more access to the healthcare system, or just are more cautious and sensitive about their own symptoms. So they don’t represent all the other people who are less likely to go to the hospital. So it was really hard during COVID to get an accurate assessment of COVID in the general population. One study I saw tested all pregnant women who came to the hospital to deliver their babies, assuming that almost every pregnant women would go to the hospital to deliver. They argued that this generated a more random sampling of the public than other studies.
Observational studies can be very useful, but because they can’t control all the conditions of the study, they can still lead to wrong or insufficient conclusions. Because they are much less expensive to run than controlled trials, they care still commonly used in the medical community.
Large, controlled, double-blinded trials: These are the gold standard of medical studies. Researchers start by defining a problem, and then collecting a lot of patients and doctors to be involved in the study. Great care is taken to select the right patients under the right set of conditions. These studies often involve hundreds to thousands of patients. Drug companies and regulators insist on every aspect of testing to be controlled. A lab next door to mine had to monitor every individual section of a freezer to make sure all of the samples, drugs, and testing materials were at the proper temperature at all times!
These studies can be very time consuming, taking months, years, and even decades, and can be very expensive. Unfortunately, many regulatory agencies will accept nothing less that this kind of study as proof of the effectiveness of a treatment. So when your doctor says “there’s no evidence that this treatment will work”, he’s just saying that a large, controlled, double-blinded trial hasn’t been done yet! And since many small companies can’t afford these studies, it can be very difficult for their treatment to be recognized as useful.
What does “double blinded” mean? If a patient is enrolled in a trial for a new cancer trial, testing a new chemotherapy agent, they will be placed in a test group or a control group. The test group is given the agent, while the control group is taken into the same medical setting, but just given a salt solution or other inert substance. This is done because the “placebo effect” is absolutely real. Many people improve in their condition after receiving fake treatments. For a new treatment to be considered useful, it has to show that it delivers more improvement than the fake treatment!
It’s important for the patients to not know what group they’re in for the conclusions to be valid. This is a single-blinded study. It’s also true that researchers, even subconsciously, can fudge data in favor of the conclusion they expect. So many don’t find the study useful unless the researcher are blinded as well. How is this done? When patients are enrolled, some researchers keep a list of patients and which group they are in. Technicians will give the treatments and give the data to a second set of researchers who will analyze the data for which patients had the best results. Finally, the test results will be compared to the list of patients and the test or control groups they were originally divided into. This way, the researchers who analyzed the data don’t know who the test patients were until the very end of the study. This way, the very “cleanest” information can be derived from the study. Obviously, this can be a very complicated process and contributes to the high cost and complexity of these trials.
I hope you found that useful!
Don’t fear, but be smart!
Erik
If you want to subscribe to these posts, click on the “Subscribe” link on the lower right corner of this page. The link will also appear if you start scrolling up from somewhere in the middle of the page.
You can read all of my cancer updates here.
