In the past few weeks in San Diego, I’ve heard several stories that discuss Ct values in regards to COVID testing. Since this is my field, I thought I’d talk about what a Ct value is and it’s relation to your results. This post is going to be pretty in the weeds, so if your not interested in the detail, you can skip this one.
PCR: The Polymerase Chain Reaction (PCR) was invented in 1983, and by the 90s, it has become a commonly used technique foundational to several molecular biology techniques, including DNA sequencing, DNA manipulation, sequence detection, and many more. Basically, the technique is used to make many many copies of a small amount a DNA. The DNA molecule is double stranded, the 2 strands are reverse copies of each other, binding to each other with weak interactions.

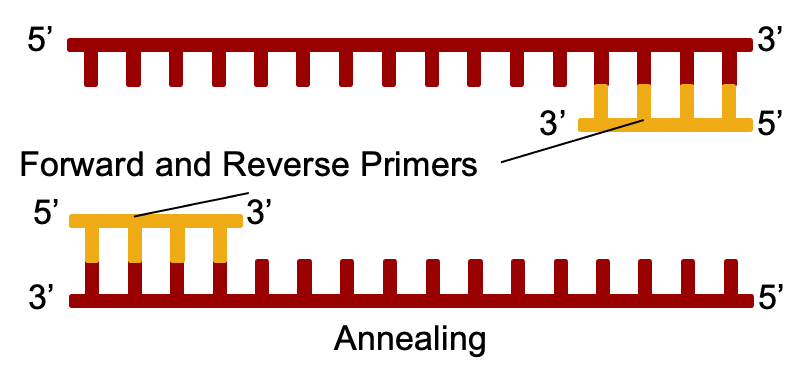
Heat is used to separate the 2 strands, and small pieces of DNA called “primers” bind to the DNA copies at a lower temperature.

The primers are designed to perfectly match sequences in the template strands. This is why PCR reactions can be very specific to a particular target, like SARS-2.
Next, the template strands are copied by a protein called “DNA Polymerase”.
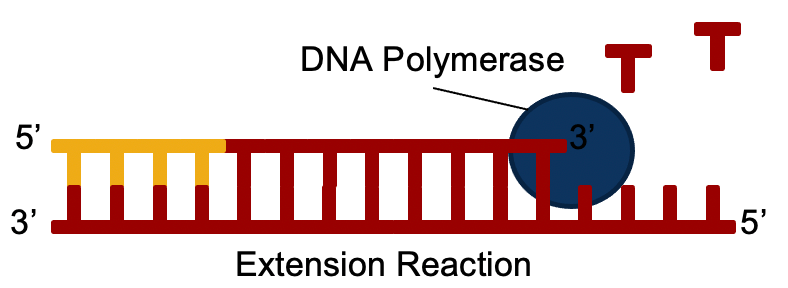
After this, the reaction is heated up again, and the process is repeated. For a PCR reaction used for detection, this is repeated 40 – 45 times. With every repetition of this process, the numbers of molecules doubles, so from every 1 molecule of starting DNA, you could theoretically end up with almost a trillion copies!
Real-time PCR: Lots of copies of DNA aren’t enough to detect it. You also need something else. Medical detection uses a process called “real-time PCR”. In this process, a third piece of DNA called a “probe” is also added. The probe has a fluorescent molecule called a “reporter” on the front end, and a molecule called a “quencher” on the back. The reporter gives off light during the reaction. The quencher is a molecule that absorbs light and coverts it to heat, effectively dimming the light coming from the reporter.
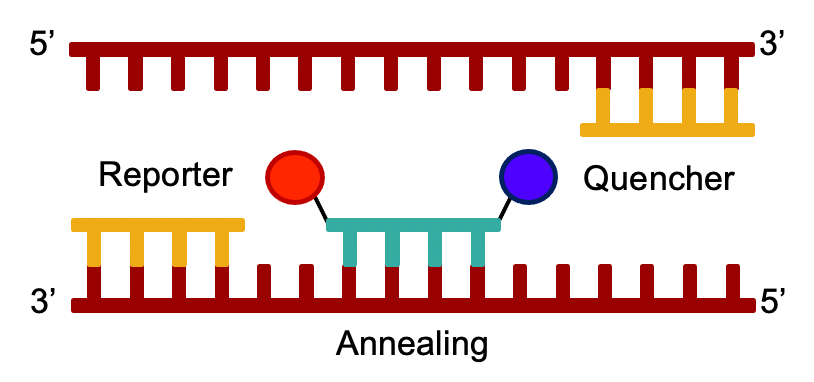
As the real-time PCR reaction progresses, the DNA Polymerase chops up the probe as well. When this happens, the quencher is separated from the reporter, and the reporter appears to give off more light! The medical instrument detects this extra light which leads to the result.

Ct values: You may have heard the term “Ct value” thrown around. As I mentioned before, with every cycle of PCR, the number of DNA copies doubles. At the same time, the reporter molecules start to give off more light. Even with all the reporter molecules around, the instrument can’t detect it until at least cycle 15. When it does, a graph of fluorescence coming from the reaction will start to show an increase.
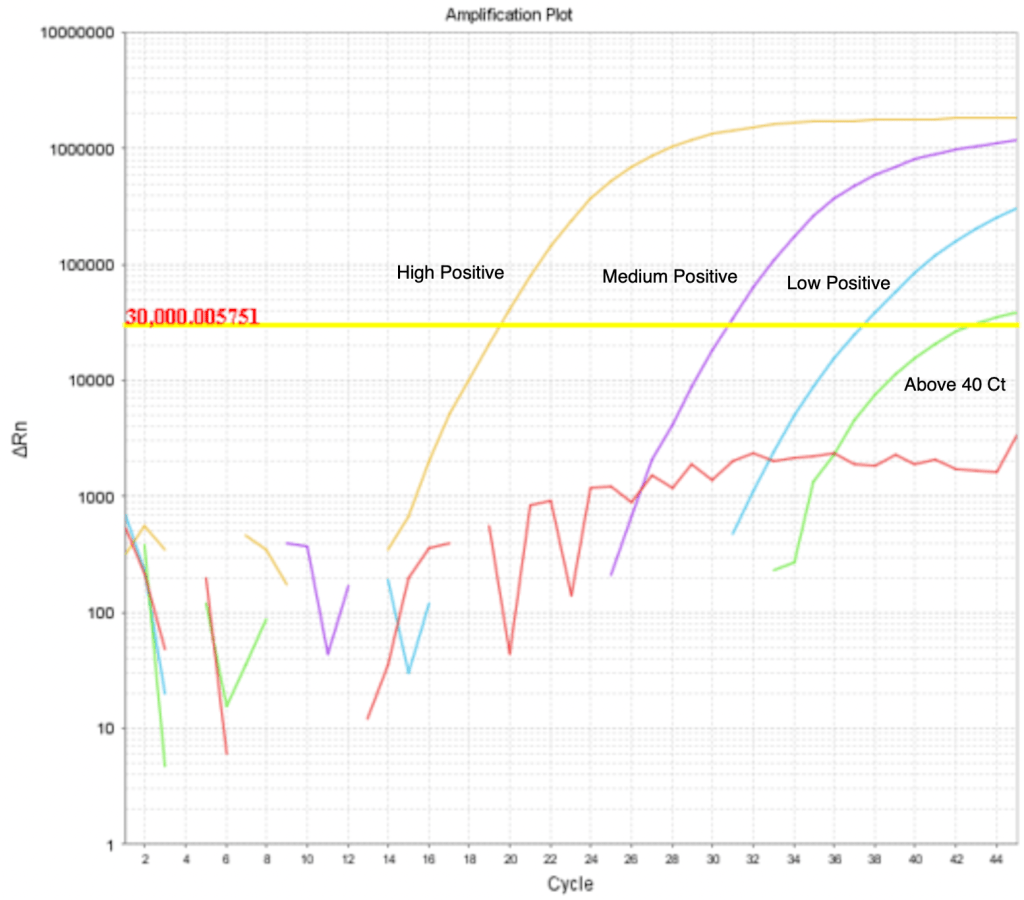
The more starting DNA you have in the reaction, the sooner the instrument will detect a rise in light. Scientists designing the test set a Cycle Threshold (the yellow line in “Real-time PCR Results”). This line is somewhat arbitrary at first, but when the test is validated, it is “set in stone” before being submitted to the FDA for approval. After the threshold line is set, the cycle at which the line of fluorescence for sample crosses the threshold line is called the “Ct value”. As you can see in the graph, the more starting DNA you have, the lower the Ct value is. The lower the starting DNA you have, the later the line will cross the Threshold, and the higher the Ct value.
A patient with a lot of SARS-2 in their sample will give a very low Ct value, almost never lower than 15-19. In the example above, the orange line represents a patient with a lot of virus. The higher the Ct value, the less virus a patient has in their sample. A sample that gives Ct value in the high 30s has very little virus, and is most likely not symptomatic. In fact, some scientists have even said that a Ct value of higher than 35 means the test is really just detecting viral debris after the virus has been cleared and the infection is basically over. A good test can detect as few as 50 virus molecules in a sample.
Most labs don’t even bother to report any result with Ct over 40. I’ve never heard of a lab reporting a result with a Ct over 45. Results like this are generally considered un-reliable, since PCR can give false positive results at very high cycle numbers. Most labs eliminate this possibility by just not reporting Cts over 40. A few weeks ago, a person at a San Diego County meeting claimed that many labs are reporting Cts over 45, and thus giving false positive results. I happen to know this man personally. We disagree on the proper approach to COVID, but he’s a good guy, and I like him personally. He is not a scientist. Anyway, I contacted him to ask him for evidence that labs are reporting Cts over 45, and I have not heard back. As I said before, I’ve never heard of a lab reporting a positive result for a real-time PCR test with a Ct over 45. So I’d be surprised if this was happening. If you have evidence of this, please let me know!
A local radio commentator in San Diego suggested on air that labs should report the Ct number. I’m all for this, but I know first hand that labs usually do not report the Ct number. In fact, many patients, and yes, even many physicians, don’t know what this number means and don’t actually want to see it in a report! Yes, that’s right, on one complicated test I built in which I included the Ct value in the report, doctors called to ask us to remove it! They said it was confusing the issue for them. This may have been because it was confusing their patients, but suffice it to say, many downstream users don’t want the Ct value and that’s why it’s not included. Generally, labs just report “COVID Positive” or “COVID Negative”. In some cases, “Detected” or “Not Detected” are used instead, to avoid confusion.
This is to avoid the issue of a patient saying “My result is positive! That’s great!” No, sir, it’s not that kind of positive.
I actually think the Ct number is very useful, and would love to see it included, but it probably won’t be.
Anyway, hope that was helpful. Your questions below will help me make this all clearer.
Don’t fear, but be smart!
Erik

2 thoughts on “PCR and the Ct Value”